A major UK study looking at whether Covid-19 vaccines can be safely mixed for the first and second doses is being expanded to include two additional jabs.
Adults over 50 who have had a first dose of Pfizer/BioNtech or Oxford/AstraZeneca are being invited to take part in the Com-Cov study, with the second dose either the same as the first one, or a shot of Moderna or Novavax.
– Why are scientists studying mixing doses?
Researchers want to determine whether combining vaccines might offer stronger, longer-lasting immunity against coronavirus and its variants.
If proven to be successful, mixing and matching doses would offer more flexibility to the UK’s national vaccine rollout programme.
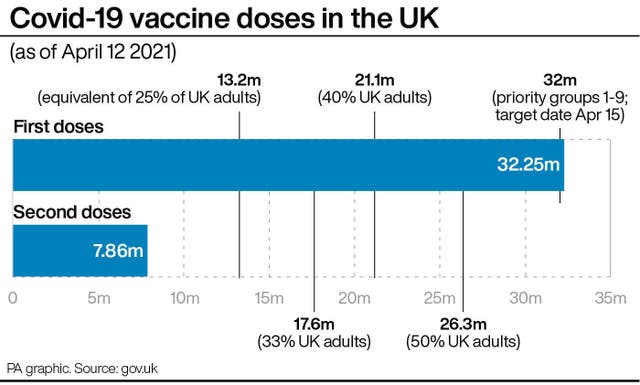
We've reached another significant milestone in our national effort – 40 million vaccine doses have been administered across the UK.
Huge thanks to the whole team involved. pic.twitter.com/XlVCynsJeQ
— Matt Hancock (@MattHancock) April 13, 2021
– How many people have been vaccinated so far?
Around 32.2 million people in the UK have had a first dose of a Covid vaccine, while 7.2 million have received a second dose.
More than 32 million people have had the first dose of their #COVID19 vaccine.
If you are aged 45 and over, you can now book your vaccine appointment.
Find out more: https://t.co/TbhWFmdpnP pic.twitter.com/KCLTj7nwYo
— Department of Health and Social Care (@DHSCgovuk) April 13, 2021
– Who is being offered the vaccine now?
In England, people aged 45 and over are being invited and can book online, while over-50s are being offered the vaccine in Scotland.
In Northern Ireland, people aged 40-45 are now eligible to book for a Covid jab.
Meanwhile, the Welsh government has said all over-50s have now been offered their first dose.
Across the UK, those aged over 16 who live with adults with weakened immune systems will also be offered a jab.
– Who is next in line to get the vaccine?
The Government has said all adults will be offered their first dose by the end of July.
This will happen in decreasing age order – starting with those aged between 40-49 years, followed by those aged 30-39 and then those in the 18-29 age bracket.
– Which vaccines are currently being used in the UK?
There are currently three vaccines approved for use in the UK: Pfizer, AstraZeneca and Moderna.
The Moderna jab works in a similar way to the Pfizer one, using tiny fragments of genetic code from coronavirus to train the body to fight off infection.
The AstraZeneca vaccine is slightly different and uses a harmless, modified virus which carries instructions that also teaches the body to tackle Covid-19.
The Novavax jab has not been approved yet in the UK, but that is expected to happen soon – with early results from trials showing it to be safe and effective.
– How many vaccine doses are there?
The UK has ordered seven vaccines involving 407 million doses – more than enough for every adult in the country.
These include AstraZeneca (100 million), Pfizer (40 million), Valneva (100 million), Novavax (60 million), GlaxoSmithKline (60 million), Janssen (30 million) and Moderna (17 million).
– Are blood clots as possible vaccine side-effects a cause for concern?
There have been reports of a rare type of bloods clot connected to the AstraZeneca vaccine.
However, the UK medicines regulator – the Medicines and Healthcare products Regulatory Agency (MHRA) – has said the benefits of the AstraZeneca vaccine continue to far outweigh any risks.
But they added that as a precautionary measure, under-30s will be offered an alternative jab.
Up to March 31, the MHRA has received 79 reports of blood clots accompanied by low blood platelet (cells that can help blood clot) count, all in people who had their first dose of the vaccine.
Of these 79, a total of 19 people have died, and three were under the age of 30, although it has not been established what the cause was in every case.
– What about the Pfizer/BioNTech or Moderna jabs?
The MHRA has said up to March 31, it had received “two reports of blood clots (thromboembolism) reported with thrombocytopenia for the Pfizer/BioNTech vaccine”.
By this date, approximately 11 million first doses and 3.5 million second doses of the vaccine had been given.
There is no data on the Moderna vaccine as it is not yet being used widely in the UK.



Comments: Our rules
We want our comments to be a lively and valuable part of our community - a place where readers can debate and engage with the most important local issues. The ability to comment on our stories is a privilege, not a right, however, and that privilege may be withdrawn if it is abused or misused.
Please report any comments that break our rules.
Read the rules here